Retatrutide
🔍 Description:
Retatrutide is a synthetic peptide designed to mimic the effects of multiple hormones, including glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon. These hormones play a crucial role in regulating weight, blood sugar, and appetite. Administered via injection, typically once a week, Retatrutide works by binding to receptors on liver cells, initiating a cascade of events that help regulate these essential metabolic functions.
💪 Health Benefits:
– Weight Loss/Fat Loss: Retatrutide has demonstrated significant efficacy in promoting weight loss, with clinical trial participants losing an average of 17.5% of their body weight.
– Blood Sugar Control: Improves long-term blood sugar control, as evidenced by lower HbA1c levels in clinical trials.
– Reduced Risk of Cardiovascular Events: May help lower the risk of heart attack and stroke by improving blood pressure, cholesterol levels, and other cardiovascular risk factors.
– Additional Potential Benefits: Retatrutide is being investigated for its potential to treat conditions such as obstructive sleep apnea and knee osteoarthritis.
💉 Dosing/Administration:
– Dosage: Clinical trials have explored doses of 1 mg, 4 mg, 8 mg, and 12 mg per week, but the optimal dosing regimen is still under investigation.
– Administration Method: Typically administered via subcutaneous injection once a week.
– Treatment Duration: As it is still under investigation, the long-term regimen will be determined by ongoing clinical trials.
⚠️ Side Effects and Safety:
– Common Side Effects: Nausea, vomiting, diarrhea, and abdominal pain, which are usually mild and temporary.
– Safety Profile: Retatrutide has been generally well-tolerated in clinical trials, but its long-term safety and efficacy are still being studied.
https://www.nature.com/articles/s41591-024-03018-2
Retatrutide: Triple Agonist Breakthrough in Obesity and Metabolic Disease Treatment
Mechanism of Action:
🔬 Retatrutide is a synthetic peptide that acts as a triple receptor agonist, targeting glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon (GCG) receptors 🚫 GLP-1 and GIP agonism reduces appetite, delays gastric emptying, and enhances insulin secretion, while glucagon agonism increases energy expenditure and promotes hepatic fat oxidation 💪 The combination shifts substrate utilization toward fat burning, elevating resting metabolic rate without the typical metabolic slowdown from calorie restriction ⚡ Glucagon receptor activation boosts fatty acid oxidation in the liver, converting stored energy into usable fuel and preventing fat accumulation 🧬 Upstream effects on energy homeostasis allow sustained fat loss independent of hunger signals, with a half-life of 6 days enabling once-weekly dosing 🔥 Dose-dependent potency: highest at GIP (EC50: 0.0643 nM), moderate at GLP-1 (0.775 nM), and lowest at glucagon (5.79 nM), creating balanced multi-pathway activation
Body Composition Effects:
📊 Phase 2 trials in obesity showed up to 24.2% total body weight reduction at 48 weeks with 12 mg dosing, versus 2.1% with placebo 🎯 In type 2 diabetes patients, retatrutide achieved superior fat mass reduction compared to placebo and dulaglutide, with estimated 80-90% of weight loss as fat 🔥 Clinical data indicate 17.1-24.2% weight loss across doses, with waist circumference reductions of 10-19 cm, signaling substantial visceral and abdominal fat loss 📏 For a 200 lb individual at 30% body fat, this equates to 40-48 lbs lost, dropping to ~15-20% body fat over 48 weeks without mandated diet changes 💪 Lean mass preservation is enhanced by glucagon-driven energy expenditure, minimizing muscle loss to <10-15% of total weight reduction in early analyses
Fat Loss Characteristics:
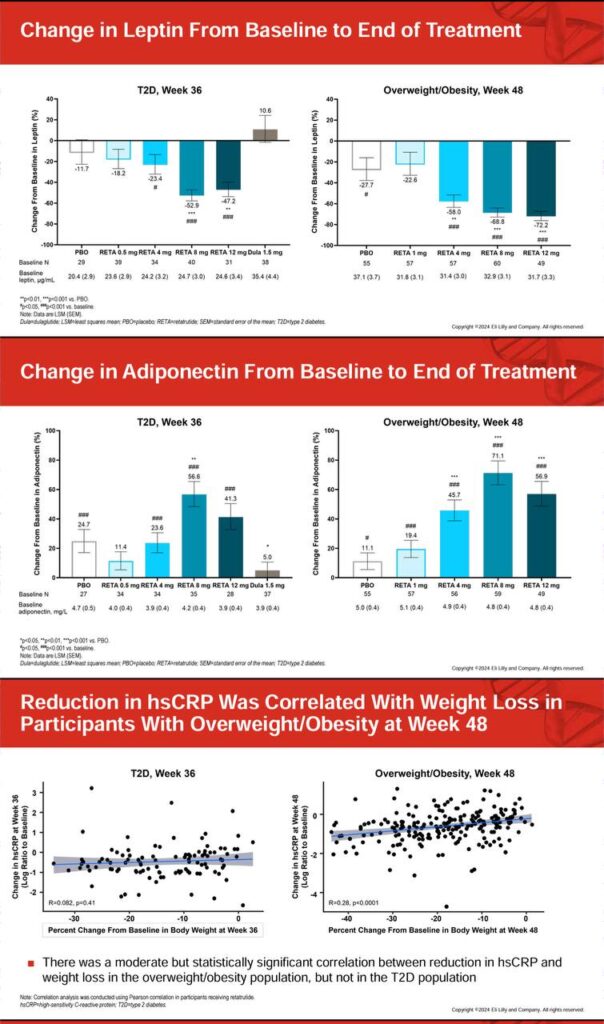
🎯 Prioritizes visceral and liver fat, with up to 86% liver fat reduction at 12 mg, achieving normal levels (<5%) in 93% of participants 🔍 Targets intra-abdominal and subcutaneous fat more effectively than dual agonists, yielding visible contouring beyond simple scale weight drops ✨ Liver fat reductions occur rapidly (within 24 weeks), independent of full weight loss, providing rapid cardiometabolic benefits
Metabolic Advantages:
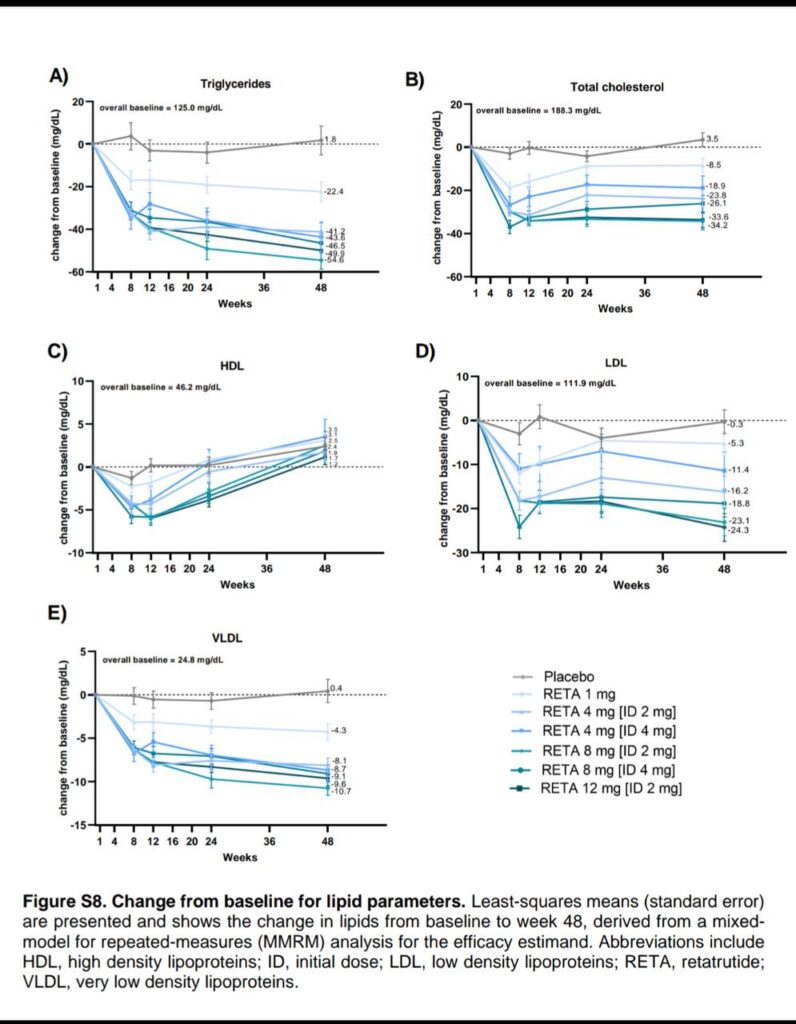
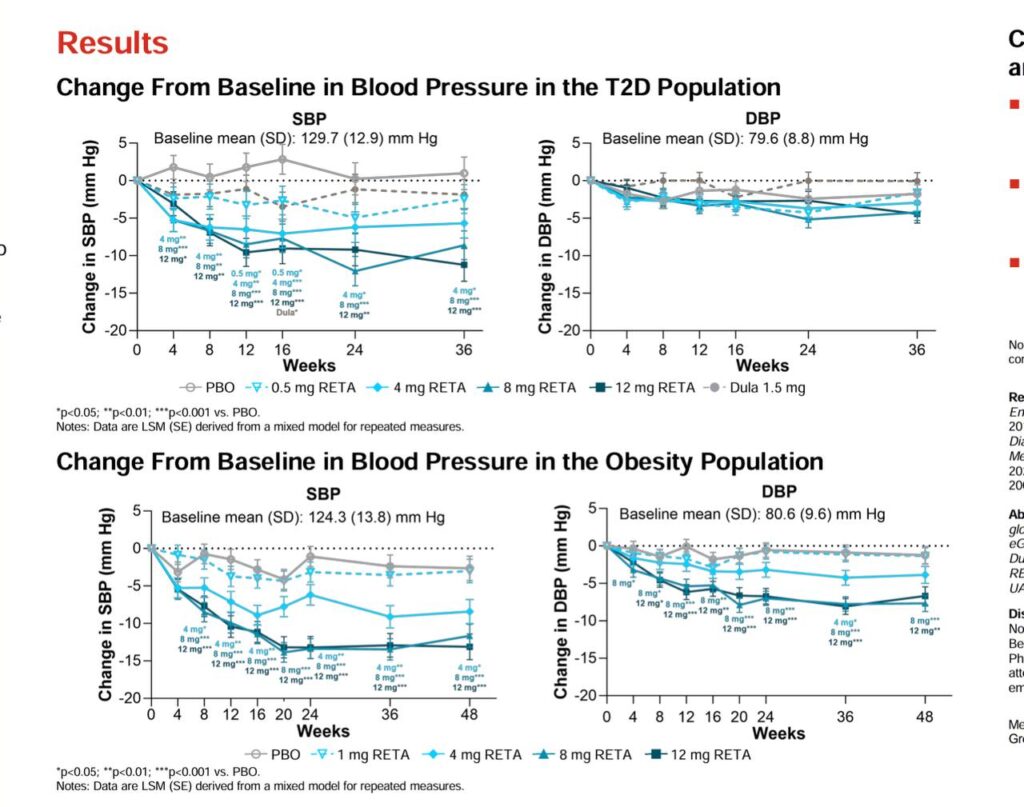
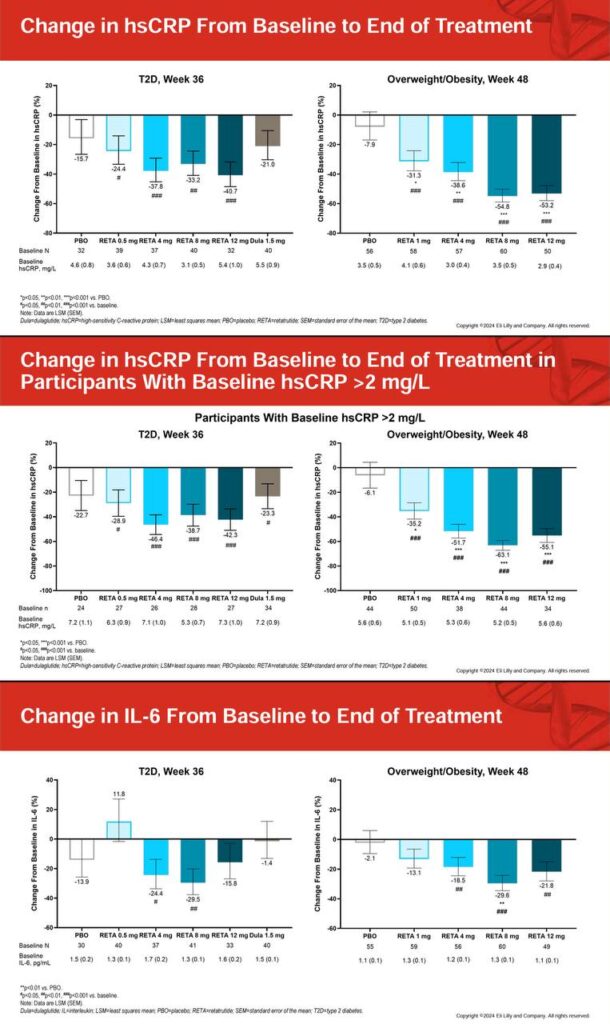
🛡️ Sustained increases in energy expenditure via glucagon, countering adaptive thermogenesis seen in caloric deficits 🔄 Improves HbA1c by up to 2.02%, fasting glucose by 23.5 mg/dL, and lipids (reduced triglycerides and LDL) ⚖️ No significant hypoglycemia risk; enhances insulin sensitivity without rebound hunger 🎯 Lowers systolic/diastolic blood pressure by 9.9/3.9 mmHg, supporting cardiovascular health during weight loss
Clinical Drug: Retatrutide:
💉 Administered as subcutaneous self-injection once weekly 📅 Dosing starts at 1-2 mg, escalating every 4 weeks to maintenance of 4-12 mg over 12 weeks (e.g., 2 mg → 4 mg → 8 mg → 12 mg) ⏱️ Full effects build over 24-48 weeks; ongoing phase 3 trials assess long-term use up to 80 weeks 🏥 Investigational; hepatic metabolism with no CYP450 interactions; half-life supports convenient weekly regimen
Expected Results with Training:
🏋️ When combined with resistance training and protein-rich diet in a mild deficit, anticipate 1-2 lbs weekly fat loss plus 0.5-1 lb lean mass gain over 12 weeks 💪 Glucagon boost amplifies training adaptations, enhancing muscle protein synthesis and recovery without fatigue 🔥 Adds 200-400 extra calories burned daily via elevated metabolism, accelerating recomp while preserving energy for workouts ✨ Users report improved endurance and motivation, yielding 25-30% total fat reduction with visible muscle definition in 6 months
…Side Effects and Management:
⚠️ Predominantly gastrointestinal (GI), mild-moderate, and transient during dose escalation 🤒 Common: nausea (up to 40% at high doses), vomiting (20-30%), diarrhea/constipation (15-25%), decreasing appetite 💉 Injection site reactions mild; heart rate increases 5-10 bpm (peaks at 24 weeks, resolves) 🩸 Rare: headache, abdominal pain; no hepatotoxicity or hypoglycemia signals 👃 Serious events ~4%, similar to placebo; discontinuation 6-16% (dose-related) ✅ Favorable profile vs. dual agonists, with lower severe GI incidence
Side Effect Management Strategies:
🔄 Slow escalation (start at 2 mg) reduces GI onset by 50%; anti-nausea meds like ondansetron for first weeks 🩺 Monitor heart rate quarterly; hydrate and electrolytes for GI symptoms 💉 Rotate sites; eat small, frequent meals to mitigate appetite suppression 🔬 Baseline/periodic labs for lipids, glucose; pause if severe GI persists >1 week 🎨 No cosmetic issues noted; symptoms self-resolve post-escalation
Combination with GLP-1 Drugs:
🔬 Phase 3 trials explore retatrutide in T2D and CVD alongside lifestyle interventions, but no direct combos yet 📊 Preclinical data suggest synergy with SGLT2 inhibitors for enhanced kidney/liver protection 💯 Future pipelines include retatrutide-like triples with amylin analogs for amplified appetite control 🛡️ Glucagon component may offset muscle loss from pure GLP-1s like semaglutide by 20-30% 📉 Potential with statins for lipid synergy; trials recruiting for OSA/CVD combos 💊 As a standalone triple, it inherently combines GLP-1/GIP benefits with glucagon edge
Synergistic Mechanism:
🍽️ GLP-1/GIP curb intake; glucagon ramps expenditure and fat oxidation 🔒 Triple action creates caloric deficit without perceived restriction, sustaining adherence ⚡ Yields 20-25% fat-specific loss, preserving lean mass better than duals 🎯 Ideal for plateaus: glucagon revives stalled metabolism during long-term use
Future Developments:
💊 Next-gen oral formulations in preclinical, targeting 2030 release 📅 Phase 3 TRIUMPH trials complete 2026, covering obesity, T2D, MASH, CVD, OSA 🎯 Wearable integrations for dose auto-adjust based on activity/glucose 🤖 AI-optimized escalation protocols to personalize based on response 🧬 Gene therapy adjuncts for receptor sensitization in late 2030s 🔮 Expanded indications: chronic pain, osteoarthritis relief via weight offload 📅 AI accelerating trial analysis for faster iterations
Commercial Availability Timeline:
✅ Phase 3 data readout mid-2025 to early 2026 📅 FDA filing anticipated late 2026 if safety holds 💉 Auto-injector pens for home use standard ⏰ Approval projected 2027; initial launch for obesity/T2D
Therapeutic Applications:
🏥 Core: obesity, overweight with comorbidities (T2D, hypertension) ⚙️ MASH reversal (86% liver fat drop); knee OA pain relief via 20%+ weight loss 🎯 CVD risk reduction; OSA symptom alleviation 💪 Muscle-sparing weight management for sarcopenic obese 🏃 Broader metabolic syndrome, including NAFLD progression halt
Ethical and Practical Considerations:
⚖️ Enhances, doesn’t replace, balanced nutrition and movement 🎯 Foundation: 80/20 diet/training split; drug as accelerator for stalled progress 💰 Misuse without basics amplifies sides, wastes resources ✨ Integrated approach yields sustainable 20%+ transformations with vitality 🔮 By 2030, triples like retatrutide normalize as first-line for metabolic health 🚀 Enables elite body comp without extremes, democratizing peak performance 📚 Discern validated science from hype; consult pros for personalization
Key Advantages Over Current Drugs:
✅ Superior 24% weight loss vs. 15-20% from tirzepatide/semaglutide ✅ Glucagon adds energy expenditure/liver fat targeting absent in duals ✅ Better lean mass retention via metabolic boost ✅ Weekly dosing with slower escalation for tolerability ✅ No new safety signals; transient GI vs. chronic in some GLP-1s ✅ Multi-indication potential (MASH, CVD) beyond weight alone
Comparison with GLP-1 Drugs:
Research knowledge hub, [Oct 2, 2025 at 6:21:27 AM]:
❌ Dual GLP-1/GIP (tirzepatide) max ~22% loss; less liver-specific ❌ Semaglutide causes more persistent GI, higher discontinuation ❌ Pure GLP-1s lack glucagon’s expenditure increase, risking more lean loss ✅ Retatrutide triples mechanisms for deeper, faster fat targeting ✅ Reduced adaptive slowdown; glucagon counters muscle catabolism ✅ Combo potential amplifies all benefits without additive sides
Surprising Insights:
🤯 24% weight loss in 48 weeks rivals bariatric surgery, without invasiveness 🔬 Glucagon flips liver from fat storage to burn mode, slashing 86% fat in months 🎯 93% normalize liver fat—outsized NASH reversal beyond weight drop 💉 Half-life enables “set-it-and-forget-it” weekly shots vs. daily hassles 🧠 Triple agonism bypasses single-pathway plateaus, sustaining loss indefinitely 🔄 Energy expenditure rises despite deficit, defying metabolic adaptation norms 📱 Future AI dosing could tailor to real-time biofeedback for zero sides 🧬 Preclinical reversals of steatohepatitis hint at cure-level metabolic resets
